Dog Meat Trade & Ebola.
International Animal Rescue Foundation Africa has been researching in depth the dog and cat meat trade all over Africa since 2012.
Worryingly news is now emerging that the Ebola virus a killer has been noted within the African states listed herein of which there is an active bushmeat trade and pet trade. Furthermore there is also decreased levels of wildlife within these areas compared to that of Southern Africa and East Africa.
Please view the research below that is related to dogs and Ebola. Research was conducted by Centers for Disease Control and Prevention
During the 2001–2002 outbreak in Gabon, we observed that several dogs were highly exposed to Ebola virus by eating infected dead animals. To examine whether these animals became infected with Ebola virus, we sampled 439 dogs and screened them by Ebola virus–specific immunoglobulin (Ig) G assay, antigen detection, and viral polymerase chain reaction amplification. Seven (8.9%) of 79 samples from the 2 main towns, 15 (15.2%) of 14 the 99 samples from Mekambo, and 40 (25.2%) of 159 samples from villages in the Ebola virus–epidemic area had detectable Ebola virus–IgG, compared to only 2 (2%) of 102 samples from France. Among dogs from villages with both infected animal carcasses and human cases, seroprevalence was 31.8%. A significant positive direct association existed between seroprevalence and the distances to the Ebola virus–epidemic area. This study suggests that dogs can be infected by Ebola virus and that the putative infection is asymptomatic.
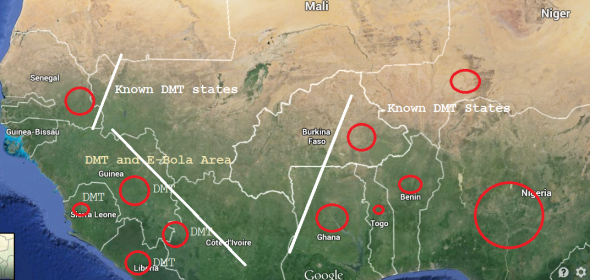
Picture (1) depicts areas where Ebola has been documented and states of Africa that have a known dog and bush meat trade. Areas too where dog and cat meat is consumed of which have yet to see any documented evidence of Ebola as yet. As you can see all the above African states are widely known to consume dog meat of which Nigeria is the largest trafficker and consumer nation. Is it not much of a coincidence that these are the only areas where dogs and cats are consumed of which Ebola has broken out. Nigeria, Mali, Ghana and Senegal have yet to document on any such cases. If this is not a coincidence then we leave you with the question, where has Ebola really derived from? No known cases have been reported in East Africa or Southern Africa to date.
During the past 3 years, 5 Ebola outbreaks due to Ebola virus-Z have struck the region of central Africa, including Gabon and Republic of Congo, and caused 334 deaths among the 428 reported human cases. In previous studies, it was showed that each extended outbreak could be subdivided into several independent epidemic clusters or chains of transmission, which resulted from close contact with an Ebola virus-Z–infected animal carcass. Epidemiologic observations and genetic analyses identified gorilla, chimpanzee, and duiker carcasses as the main sources of human cases. It must be noted too that stray dogs will consume dead wildlife meat that could be infected with Ebola. Once the species barrier has been crossed between animals and humans, the disease spreads among humans by direct physical contact.
Some human cases in the recent outbreak in the Gabon/Republic of Congo region did not have a documented source of exposure to Ebola hemorrhagic fever. Similarly, 14 (4.9%) of the 284 cases in the 1976 Sudan outbreak (6) and 55 (17.4%) of the 316 cases during the 1995 outbreak in Kikwit (7), Democratic Republic of Congo (DRC, former Zaire), had no direct physical contact with an infected person or known infected carcass. These observations point to other routes of transmission (e.g., human-human respiratory tract infection through droplets and aerosols) or may suggest that other, unidentified animal sources may be involved in Ebola virus transmission to humans.
Ebola hemorrhagic fever outbreaks occurred in villages where people keep domestic animals, including dogs. The dogs are not fed and have to scavenge for their food. They eat small dead animals found near the villages and also internal organs of wild animals hunted and slaughtered by villagers. Some dogs are also used for hunting in the dense forested area. Although canine infection by Ebola virus has never been documented, domestic dogs’ behavior and diet place them at risk.
Scientists examined whether pet dogs could have been infected by Ebola virus and their potential role as primary or secondary sources of human infection. Researchers conducted a large-scale serologic survey to determine the prevalence of Ebola virus infection in pet dogs in an Ebola virus–epidemic area of Gabon.
Sampling was conducted in 2 ways. 1) Dogs in Libreville and Port Gentil were sampled in a veterinary clinic. Blood was collected in 5-mL dry Vacutainers (VWR International, Fontenay-sous-bois, France), and serum was prepared by centrifugation. Serum specimens were stored at –20°C until they were sent to the Centre International de Recherches Médicales de Franceville (CIRMF), Gabon, where they were stored at –80°C until testing. 2) Dogs from the Ebola virus–endemic area were sampled in the villages. An experienced veterinary team was located at Mekambo, where field laboratory facilities were set up; blood samples were collected on a daily basis in the vicinity of the village by using 5-mL dry Vacutainers and medetomidine anesthesia. The tubes were then transported to Mekambo each evening, and serum was decanted from whole blood. Serum samples were kept in liquid nitrogen in 1-mL aliquots at Mekambo until they were transported to CIRMF. Serum samples were then stored at –80°C until serologic testing, antigen detection, and RNA amplification were carried out.
Dog owners were interviewed on their pets’ activities (e.g., participation in hunting) and health history. The focus of the interviews was on potential Ebola virus–exposure events, including human cases that occurred in the village and among dog owners.
Laboratory Investigations
Ebola virus–specific immunoglobulin (Ig) G was detected by using a standard enzyme-linked immunosorbent assay (ELISA) method as previously described (8). Briefly, Maxisorp plates (VWR International) were coated with Ebola virus–Z antigens diluted 1:1,000 in phosphate-buffered saline (PBS), overnight at 4°C. Control plates were coated with uninfected Vero cell culture antigens in the same conditions. Sera diluted 1:400 in 5% nonfat milk in PBS-Tween 20 (0.1%) were added to the wells and incubated overnight at 4°C. IgG binding was visualized by using a peroxidase-labeled anti-dog IgG (Kirkegaard & Perry Laboratories, Inc., Gaithersburg, MD, USA) and the TMB detector system (Dynex Technologies, Issy-les-Moulineaux, France). Optical density (OD) was measured at 450 nm with an ELISA plate reader. For each sample we calculated the corrected OD as the OD of the antigen-coated well minus the OD of the corresponding control well. The cut-off value (CO) was calculated as follows: CO = M + 3σ, where M is the average of the corrected OD of the 102 negative controls from France, and σ is the standard deviation. Samples were considered positive when the corrected OD was above the cut-off.
Samples positive in these serologic assays were used for antigen detection (9) and for viral polymerase chain reaction (PCR) amplification (10). Three positive and 3 negative serum specimens were also used for virus isolation (9). Briefly, Maxisorp plates were coated with a cocktail of 7 monoclonal antibodies against Ebola virus–Z antigens; control plates were coated with normal mouse ascitic fluid produced from the parent myeloma cell line. Serum was then added to the wells, followed by hyperimmune rabbit Ebola polyvalent antiserum and then peroxidase-conjugated goat antibodies against rabbit IgG. The TMB Microwell peroxidase substrate system was used to measure OD. For the detection of viral mRNA, total RNA was isolated from serum with the QIAmp viral RNA kit (Qiagen, Courtaboeuf, France), and cDNA was synthesized from mRNA as previously described. Two pairs of degenerate primers corresponding to the L-gene of Ebola virus were used for 2 rounds of amplification, which yielded a 298-bp fragment.
Statistical Methods
Confidence intervals for proportions were calculated by using the Clopper and Pearson method (11). Statistical comparisons between seroprevalence rates according to the sampling area were performed by using the Fisher exact test. The Cochran-Armitage test was used as a trend test for proportions, after checking for the goodness-of-fit of the underlying linear model (12). All tests used a 0.05 significance level. Statistical analyses were performed by using R software (R Development Core Team; 13).
Results
A total of 439 blood samples from dogs were screened for Ebola virus–specific IgG. Two (2%) of the 102 blood samples from dogs living in France had detectable Ebola virus–reactive IgG (Table 2). Seven of the 79 dogs sampled in Libreville and Port Gentil (8.9% prevalence rate), 15 of the 99 dogs sampled in Mekambo (15.2% prevalence rate), and 40 of the 159 dogs sampled in villages located within the Ebola virus–epidemic area (25.2% prevalence rate) had detectable IgG to Ebola virus antigens.
During the 2001–2002 Ebola outbreak in Gabon, human cases of Ebola virus infection appeared only in certain villages within the Ebola virus–epidemic area (Figure 1). The prevalence of Ebola virus-reactive IgG among dogs from the villages where humans cases occurred was 27.2%, compared to 22.4% among dogs from villages where no human cases were noted (Table 2). In some cases, hunters had brought back to the village an Ebola virus–infected animal carcass found in the forest. This carcass was the source of human infection in the village, and the disease then spread from human to human, both within the village and to other villages by population movement. Thus, only secondary human cases were observed in some villages, with no identified animal source. The prevalence rate among dogs from villages with both an animal source and human cases was as high as 31.8%, compared to 15.4% among dogs from villages with human cases but no identified animal source.
In conclusion, this study offers the first evidence that dogs might be asymptomatically infected by Ebola virus in the wild. This finding has potential implications for preventing and controlling human outbreaks. The increasing canine seroprevalence gradient from low-risk to at-risk Ebola virus–endemic areas indicates that this seroprevalence might be used as an epidemiologic indicator of virus circulation in regions where no other means of virus detection are available.
Read the entire study here now;
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3298261/
WHO quotes nothing about the transmission of Ebola from dogs to humans which is worrying.
Ebola is introduced into the human population through close contact with the blood, secretions, organs or other bodily fluids of infected animals. In Africa, infection has been documented through the handling of infected chimpanzees, gorillas, fruit bats, monkeys, forest antelope and porcupines found ill or dead or in the rainforest.
Ebola then spreads in the community through human-to-human transmission, with infection resulting from direct contact (through broken skin or mucous membranes) with the blood, secretions, organs or other bodily fluids of infected people, and indirect contact with environments contaminated with such fluids. Burial ceremonies in which mourners have direct contact with the body of the deceased person can also play a role in the transmission of Ebola. Men who have recovered from the disease can still transmit the virus through their semen for up to 7 weeks after recovery from illness.
Health-care workers have frequently been infected while treating patients with suspected or confirmed EVD. This has occurred through close contact with patients when infection control precautions are not strictly practiced.
Among workers in contact with monkeys or pigs infected with Reston ebolavirus, several infections have been documented in people who were clinically asymptomatic. Thus, RESTV appears less capable of causing disease in humans than other Ebola species.
However, the only available evidence available comes from healthy adult males. It would be premature to extrapolate the health effects of the virus to all population groups, such as immuno-compromised persons, persons with underlying medical conditions, pregnant women and children. More studies of RESTV are needed before definitive conclusions can be drawn about the pathogenicity and virulence of this virus in humans.
Signs and symptoms
EVD is a severe acute viral illness often characterized by the sudden onset of fever, intense weakness, muscle pain, headache and sore throat. This is followed by vomiting, diarrhoea, rash, impaired kidney and liver function, and in some cases, both internal and external bleeding. Laboratory findings include low white blood cell and platelet counts and elevated liver enzymes.
People are infectious as long as their blood and secretions contain the virus. Ebola virus was isolated from semen 61 days after onset of illness in a man who was infected in a laboratory.
The incubation period, that is, the time interval from infection with the virus to onset of symptoms, is 2 to 21 days.
Diagnosis
Other diseases that should be ruled out before a diagnosis of EVD can be made include: malaria, typhoid fever, shigellosis, cholera, leptospirosis, plague, rickettsiosis, relapsing fever, meningitis, hepatitis and other viral haemorrhagic fevers.
Ebola virus infections can be diagnosed definitively in a laboratory through several types of tests:
- enzyme-linked immunosorbent assay (ELISA)
- antigen detection tests
- serum neutralization test
- reverse transcriptase polymerase chain reaction (RT-PCR) assay
- virus isolation by cell culture.
Samples from patients are an extreme biohazard risk; testing should be conducted under maximum biological containment conditions.
Prevention and treatment
No vaccine for EVD is available. Several vaccines are being tested, but none are available for clinical use.
Severely ill patients require intensive supportive care. Patients are frequently dehydrated and require oral rehydration with solutions containing electrolytes or intravenous fluids.
No specific treatment is available. New drug therapies are being evaluated.
Natural host of Ebola virus
In Africa, fruit bats, particularly species of the genera Hypsignathus monstrosus, Epomops franqueti and Myonycteris torquata, are considered possible natural hosts for Ebola virus. As a result, the geographic distribution of Ebolaviruses may overlap with the range of the fruit bats.
Ebola virus in animals
Although non-human primates have been a source of infection for humans, they are not thought to be the reservoir but rather an accidental host like human beings. Since 1994, Ebola outbreaks from the EBOV and TAFV species have been observed in chimpanzees and gorillas.
RESTV has caused severe EVD outbreaks in macaque monkeys (Macaca fascicularis) farmed in Philippines and detected in monkeys imported into the USA in 1989, 1990 and 1996, and in monkeys imported to Italy from Philippines in 1992.
Since 2008, RESTV viruses have been detected during several outbreaks of a deadly disease in pigs in Philippines and China. Asymptomatic infection in pigs has been reported and experimental inoculations have shown that RESTV cannot cause disease in pigs.
Prevention
Controlling Ebola Reston in domestic animals
No animal vaccine against RESTV is available. Routine cleaning and disinfection of pig or monkey farms (with sodium hypochlorite or other detergents) should be effective in inactivating the virus.
If an outbreak is suspected, the premises should be quarantined immediately. Culling of infected animals, with close supervision of burial or incineration of carcasses, may be necessary to reduce the risk of animal-to-human transmission. Restricting or banning the movement of animals from infected farms to other areas can reduce the spread of the disease.
As RESTV outbreaks in pigs and monkeys have preceded human infections, the establishment of an active animal health surveillance system to detect new cases is essential in providing early warning for veterinary and human public health authorities.
Reducing the risk of Ebola infection in people
In the absence of effective treatment and a human vaccine, raising awareness of the risk factors for Ebola infection and the protective measures individuals can take is the only way to reduce human infection and death.
In Africa, during EVD outbreaks, educational public health messages for risk reduction should focus on several factors:
- Reducing the risk of wildlife-to-human transmission from contact with infected fruit bats or monkeys/apes and the consumption of their raw meat. Animals should be handled with gloves and other appropriate protective clothing. Animal products (blood and meat) should be thoroughly cooked before consumption.
- Reducing the risk of human-to-human transmission in the community arising from direct or close contact with infected patients, particularly with their bodily fluids. Close physical contact with Ebola patients should be avoided. Gloves and appropriate personal protective equipment should be worn when taking care of ill patients at home. Regular hand washing is required after visiting patients in hospital, as well as after taking care of patients at home.
- Communities affected by Ebola should inform the population about the nature of the disease and about outbreak containment measures, including burial of the dead. People who have died from Ebola should be promptly and safely buried.
Pig farms in Africa can play a role in the amplification of infection because of the presence of fruit bats on these farms. Appropriate biosecurity measures should be in place to limit transmission. For RESTV, educational public health messages should focus on reducing the risk of pig-to-human transmission as a result of unsafe animal husbandry and slaughtering practices, and unsafe consumption of fresh blood, raw milk or animal tissue. Gloves and other appropriate protective clothing should be worn when handling sick animals or their tissues and when slaughtering animals. In regions where RESTV has been reported in pigs, all animal products (blood, meat and milk) should be thoroughly cooked before eating.
Controlling infection in health-care settings
Human-to-human transmission of the Ebola virus is primarily associated with direct or indirect contact with blood and body fluids. Transmission to health-care workers has been reported when appropriate infection control measures have not been observed.
It is not always possible to identify patients with EBV early because initial symptoms may be non-specific. For this reason, it is important that health-care workers apply standard precautions consistently with all patients – regardless of their diagnosis – in all work practices at all times. These include basic hand hygiene, respiratory hygiene, the use of personal protective equipment (according to the risk of splashes or other contact with infected materials), safe injection practices and safe burial practices.
Health-care workers caring for patients with suspected or confirmed Ebola virus should apply, in addition to standard precautions, other infection control measures to avoid any exposure to the patient’s blood and body fluids and direct unprotected contact with the possibly contaminated environment. When in close contact (within 1 metre) of patients with EBV, health-care workers should wear face protection (a face shield or a medical mask and goggles), a clean, non-sterile long-sleeved gown, and gloves (sterile gloves for some procedures).
Laboratory workers are also at risk. Samples taken from suspected human and animal Ebola cases for diagnosis should be handled by trained staff and processed in suitably equipped laboratories.
View more within the video below.
International Animal Rescue Foundation and Say No To Dog Meat.Net whom are not affiliated with us have been stating now for many months that dog and cat meat trade “will pose a significant health risk” in viral format to the global population unless the trade is ceased. Could the Ebola outbreak now be one of these major global concerns and if so why has not one single government lobbied by us and SNTDM actually taken this on board. We will most likely never know where the Ebola outbreak originated from. This is plausible evidence though that Ebola can be transmitted from dogs to humans and from dogs that have brought back infested Ebola meat to human villages. Is it such a coincidence too that Ebola has only been recorded within Western Africa where dog, cat and bush meat is consumed and where poverty, climate change, civil unrest and lack of wildlife species is common place. Only one single case of Ebola this year as been recorded within Southern Africa that we are aware of.
We have highlighted this issue in the hope that someone within the virial community will investigate in-depth the possible relations of consuming dogs and bush meat and Ebola virus documenting a detailed response to our concerns as to why Ebola has only been recorded within Western Africa. Bush meat is consumed all over Africa, yet dog meat is consumed only by small minorities with the exception of Nigeria. Coincidence or are the authorities concealing what they have actually ignored?
Whatever the answer this must be a wake up call to the entire African continent now before its to late.




Pingback: Africa, Kenya: 6 Months Jail For Dog Killer | SAY NO TO DOG MEAT.NET